|
6/18/2023 0 Comments Lateral flow antigen test 
Two additional experimental approaches were evaluated in this study–a mobile phone application to image and interpret a completed LFA, and a system to evaluate a patient’s sense of smell. Here we report the subsequent clinical validation procedures and results from this prototype lateral flow assay, in comparison to RT-qPCR results from swabs taken from nasopharyngeal and anterior nares sampling. Specifically, the analytical sensitivity achieved the target WHO target of 10 6 genomic copies/mL. In-vitro performance of the assay met the World Health Organization (WHO) target product profile (TPP) for a rapid diagnostic test. This assay is distinct from available commercial assays in that the assay design and architecture were released as open access, and that all reagents are commercially available. We previously published the development of a rapid test in the lateral flow assay (LFA) format to detect the nucleocapsid protein of the SARS-CoV-2 virus. The Food and Drug Administration (FDA) has provided Emergency Use Authorizations (EUA) to (as of ) 24 rapid antigen diagnostic tests, most of which are based on detection of SARS-COV2 nucleocapsid protein All of these assays are proprietary and are produced by commercial entities. While they may not be as sensitive as PCR, antigen-based rapid tests are generally inexpensive, amenable to mass-production, can be conducted by minimally trained individuals, and offer nearly immediate results for public health actions. Simpler and less-expensive antigen-based rapid tests have been developed to address these issues. For example, time to results may be prolonged, cold chain may be required to transfer samples to lab facilities, expensive reagents are utilized, and trained lab technicians are needed to perform the testing. 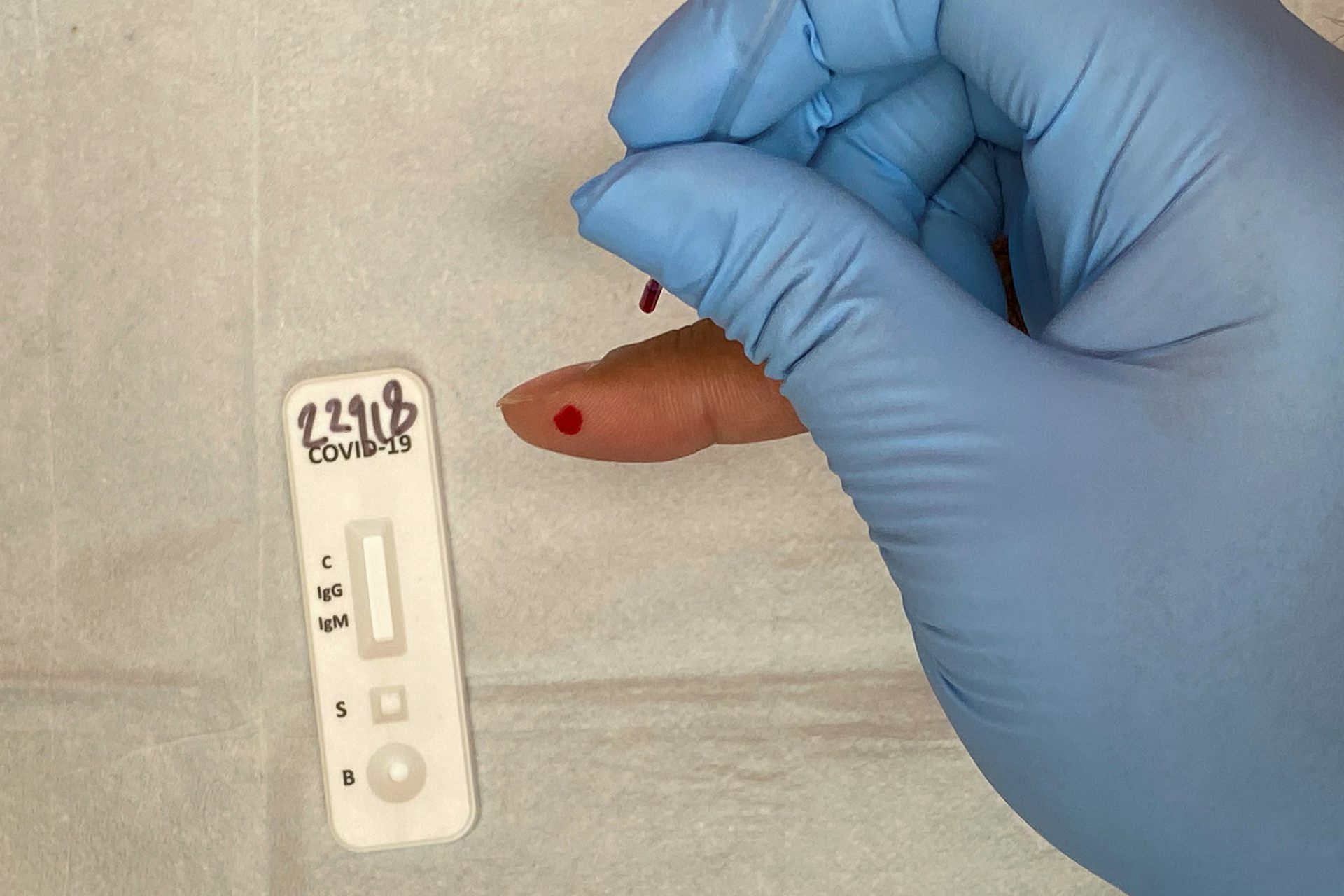
While enormously sensitive, PCR poses several challenges. Īt present, most health facilities rely on polymerase chain reaction (PCR) -based tests, which are based on amplification of genetic material for detection of the virus. Transmission models have predicted early detection in the community coupled with isolation may accelerate reductions in transmission. One of the most effective tools to detect potentially infectious cases of COVID-19 are rapid diagnostic tests for the virus that causes the disease, SARS-COV-2. With more than 3,250,000 deaths worldwide due to the Covid-19 pandemic, early detection of infectious patients is required to aid in efforts to interrupt transmission. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are within the paper and its Supporting Information files.įunding: All Authors of the manuscript work for the funding organization - Global Health Labs.Ĭompeting interests: No authors have competing interests. Received: JAccepted: AugPublished: August 17, 2021Ĭopyright: © 2021 Bachman et al. PLoS ONE 16(8):Įditor: Etsuro Ito, Waseda University: Waseda Daigaku, JAPAN (2021) Clinical validation of an open-access SARS-COV-2 antigen detection lateral flow assay, compared to commercially available assays. Citation: Bachman CM, Grant BD, Anderson CE, Alonzo LF, Garing S, Byrnes SA, et al. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
